Is This the Future of Soil Carbon Analysis?
Accurate analysis is critical for understanding soil health and quantifying soil organic carbon (SOC) and other soil properties. After a soil sample has been collected from a field enrolled in Carbon by Indigo or the Market+ Source program, it’s promptly sent to a commercial laboratory for analysis. There, the sample undergoes elemental analysis by a process known as “dry combustion.” The soil is dried, ground, sieved, and, if necessary, treated with acid to remove any inorganic carbon, and then heated at very high temperatures (>900 °C). The organic material in the sample is oxidized and the amount of CO2 emitted corresponds to the percentage of SOC.
Dry combustion produces accurate results, but it’s also relatively slow, labor intensive and expensive. As more programs focused on climate-smart agriculture grow across the industry, soil lab sample analysis could become a bottleneck. What if there was a way to expedite the process while reducing costs?
Soil Spectroscopy as an Alternative
In soil spectroscopy, light is emitted from a spectrophotometer and then either absorbed or reflected by the soil of interest. The interaction between light over a range of wavelengths and the soil is known as the soil’s spectra. The spectra, particularly in the mid-infrared (MIR) region, is a unique signature that can inform us about multiple soil properties, like texture or carbon content, without directly measuring those properties. Using soil spectroscopy, hundreds of soil samples can be analyzed within a single day, compared to a week for traditional analytical methods. Before doing MIR soil spectral analysis, however, a laborious process called fine milling is required. Although common in research facilities, it’s a step beyond the typical grinding and sieving done in commercial soil labs.
Viability Without Fine Milling?
Past research has yielded conflicting results as to whether fine milling is a necessary step to produce accurate and unbiased predictions of soil properties. Scientists at the Woodwell Climate Research Center led a recent study published in Soil Security, that tested the predictive performance of MIR spectroscopy without fine milling on varying soil properties (clay, sand, pH, bulk density, total carbon). More than 2,300 samples from Indigo’s Soil Carbon Experiment and the Soil Health Institute’s North American Project to Evaluate Soil Health
Photographs courtesy the Woodwell Climate Research Center
Measurements were used in the study to represent soils with diverse characteristics over a wide geographic extent.
Ultimately, the study found that the accuracy and precision of soil property estimates from samples that were not finely milled were equivalent to the analysis of those that had undergone fine milling.
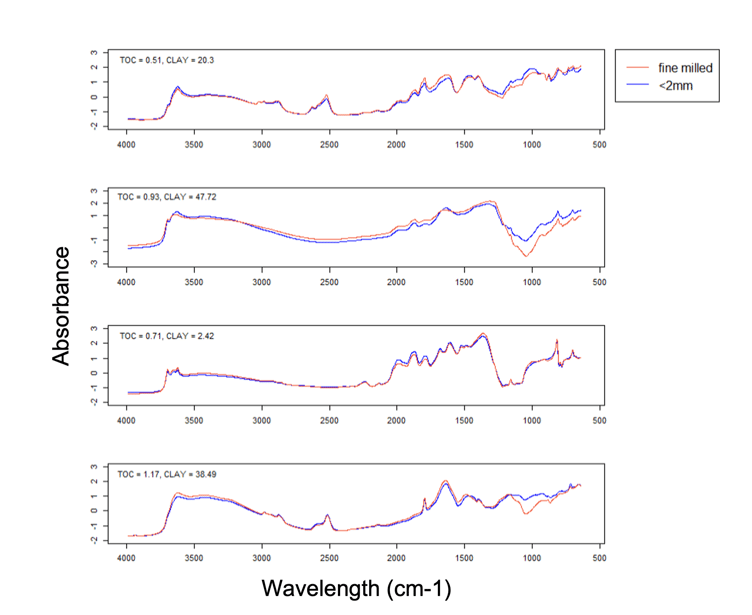
The figure above, sourced from Sanderman et al. (2023), shows an example of the spectra for four soil samples (1 per panel), each with differing amounts of clay and carbon. The blue and red lines compare finely milled and sieved results.
Implications of the Study
Results suggest that samples do not need to be finely milled when analyzing via MIR spectroscopy, which makes a faster method of analysis… even faster! Additionally, MIR spectroscopy enables greater knowledge of a variety of soil properties from just a single analysis. Determining the carbon content, texture, and pH of a soil requires just a single 60 second scan as opposed to three separate analyses, meaning more information in less time and at less cost. If the burden of fine milling is removed, then commercial laboratories may be more inclined to offer analysis of soil properties via MIR.
This research is ultimately an important step towards the commercialization of soil spectroscopy. With the increase in federal climate smart commodity programs, corporate Scope 3 programs, and carbon markets, demand for soil sample analysis will correspondingly increase. “MIR spectroscopy could help greatly increase lab throughput and affordability, making it easier to accurately monitor climate outcomes of these programs,” says Jon Sanderman, soil scientist at the Woodwell Climate Research Center and lead author of this paper.


